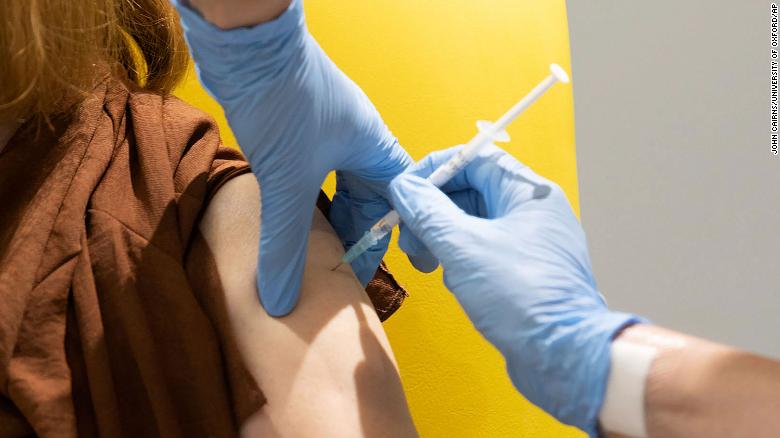
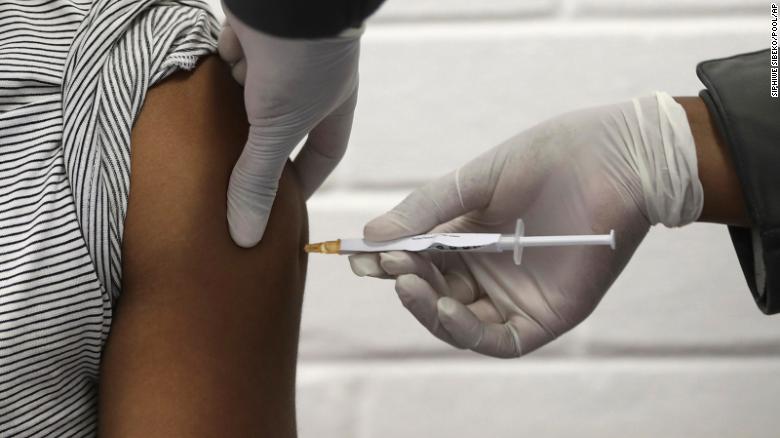
Why Oxford’s Covid-19 vaccine could do more for the world than other shots
London (CNN)In the days since Oxford University and AstraZeneca unveiled the results of the partnership’s Phase 3 Covid-19 vaccine trials, a growing number of questions have emerged.